 Learning and Development
At Sungen Biomedical, people are the most valuable resource driving innovation in therapeutic antibody development. For this reason we have built a comprehensive and forward-looking learning and development system that aims to train experts in antibody science and innovative managers at industry-leading levels. The following are the core elements of our learning and development plan:
Learning and Development
At Sungen Biomedical, people are the most valuable resource driving innovation in therapeutic antibody development. For this reason we have built a comprehensive and forward-looking learning and development system that aims to train experts in antibody science and innovative managers at industry-leading levels. The following are the core elements of our learning and development plan:
 First Show Vienna! Phase I Clinical Data of the World's First Antibody Drug SGC001 for Acute Myocardial Infarction Unveiled BIO Europe2025
On November 3, 2025, Vienna time, Sungen announced the Phase I clinical results of SGC001 Injection, the world's first antibody drug for acute myocardial infarction, at the 31st BIO Europe 2025, h...
First Show Vienna! Phase I Clinical Data of the World's First Antibody Drug SGC001 for Acute Myocardial Infarction Unveiled BIO Europe2025
On November 3, 2025, Vienna time, Sungen announced the Phase I clinical results of SGC001 Injection, the world's first antibody drug for acute myocardial infarction, at the 31st BIO Europe 2025, h...
 Home
Home
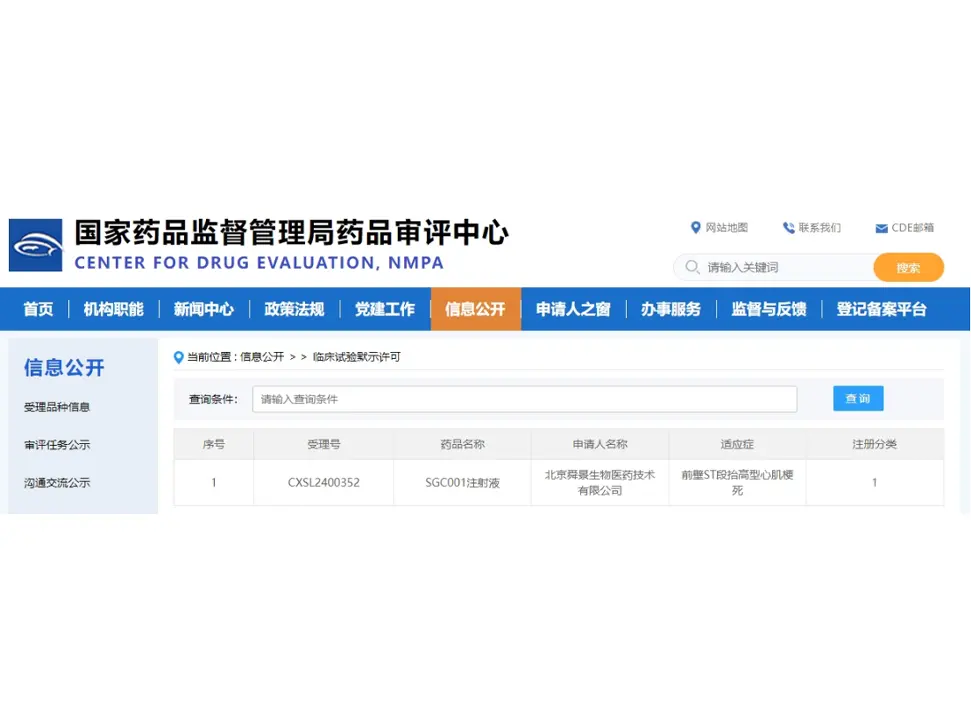 FIC! Sungen Biomedical's SGC001 Injection is Approved for Clinical Trials, Achieving Two Approvals Both in China and the United States!
On August 05, 2024, the world's first innovative drug SGC001 injection obtained the clinical trial implied approval from the Center for Drug Evaluation (CDE). The drug is developed by Beijing Sung...
FIC! Sungen Biomedical's SGC001 Injection is Approved for Clinical Trials, Achieving Two Approvals Both in China and the United States!
On August 05, 2024, the world's first innovative drug SGC001 injection obtained the clinical trial implied approval from the Center for Drug Evaluation (CDE). The drug is developed by Beijing Sung...
 Immunity
Immunity | Mast cells regulate lung inflammation through PGE2-sST2
Immunity
Immunity | Mast cells regulate lung inflammation through PGE2-sST2
 Riding the wave, The world's first acute myocardial infarction antibody SGC001 of Sungen Biomedical was presented at U.S. J.P. Morgan Conference
On January 12th, the 43rd Annual J.P. Morgan Healthcare Conference (JPM Conference), an influential investment seminar, took place in San Francisco. Sungen Biomedical made a notable presentation featu...
Riding the wave, The world's first acute myocardial infarction antibody SGC001 of Sungen Biomedical was presented at U.S. J.P. Morgan Conference
On January 12th, the 43rd Annual J.P. Morgan Healthcare Conference (JPM Conference), an influential investment seminar, took place in San Francisco. Sungen Biomedical made a notable presentation featu...
 About Us
Sungen Biomedical was founded in 2019 which is an innovative antibody drug research and development enterprise incubated by Hotgen Biotech (SH688068), a listed company on the Sci-Tech innovation board...
About Us
Sungen Biomedical was founded in 2019 which is an innovative antibody drug research and development enterprise incubated by Hotgen Biotech (SH688068), a listed company on the Sci-Tech innovation board...
 First in the World! Sungen Biomedical Obtains FDA's Clinical Trial Approval of IND SGC001 as An Emergency-use Antibody Drug for AMI
First in the world! Sungen Biomedical obtains FDA's clinical trial approval of IND SGC001 as an emergency-use antibody drug for AMIOn May 23rd, 2024, Eastern Standard Time, US Food and Drug Administr...
First in the World! Sungen Biomedical Obtains FDA's Clinical Trial Approval of IND SGC001 as An Emergency-use Antibody Drug for AMI
First in the world! Sungen Biomedical obtains FDA's clinical trial approval of IND SGC001 as an emergency-use antibody drug for AMIOn May 23rd, 2024, Eastern Standard Time, US Food and Drug Administr...
 Cell Research
Cell Research | Huaqiang Xu Collaborated with Wei Fu to Reveal the Molecular Basis of Ligand Recognition and Activation of the Human Succinate Receptor
Cell Research
Cell Research | Huaqiang Xu Collaborated with Wei Fu to Reveal the Molecular Basis of Ligand Recognition and Activation of the Human Succinate Receptor
 Sungen Biomedical's world-first new drug SGC001 monoclonal antibody receives FDA fast track approval
On March 17, 2025, US time, the world’s first acute myocardial infarction (AMI) antibody drug, SGC001, developed by Sungen Biomedical—an innovative biopharmaceutical company incubated by Beijing Hot...
Sungen Biomedical's world-first new drug SGC001 monoclonal antibody receives FDA fast track approval
On March 17, 2025, US time, the world’s first acute myocardial infarction (AMI) antibody drug, SGC001, developed by Sungen Biomedical—an innovative biopharmaceutical company incubated by Beijing Hot...
 Company Overview
We are one of the new core medical service centers under the Ukrainian Biobanking Association, the world's leading research institution for stem cells and modern biomedicine.
Company Overview
We are one of the new core medical service centers under the Ukrainian Biobanking Association, the world's leading research institution for stem cells and modern biomedicine.
 Benefits & Compensation
At Sungen Biomedical, we believe that employees are the most valuable to the company. In order to create a work environment that encourages innovation and growth, we offer a comprehensive and competit...
Benefits & Compensation
At Sungen Biomedical, we believe that employees are the most valuable to the company. In order to create a work environment that encourages innovation and growth, we offer a comprehensive and competit...
 Good News! The Laboratory Animal Management Center at Sungen Biomedical is Granted
Recently, Beijing Municipal Science and Technology Commission organized experts to conduct on-site evaluation on "Experimental Animal Use License (New)" of Sungen Biomedical Laboratory Anima...
Good News! The Laboratory Animal Management Center at Sungen Biomedical is Granted
Recently, Beijing Municipal Science and Technology Commission organized experts to conduct on-site evaluation on "Experimental Animal Use License (New)" of Sungen Biomedical Laboratory Anima...
 Nat Immunol
Nat Immunol | Minxuan Sun, Shiyang Li, and Changgeng Peng Found the Regulatory Role of NPM1 in Inflammatory Bowel Disease and Colorectal Cancer
Nat Immunol
Nat Immunol | Minxuan Sun, Shiyang Li, and Changgeng Peng Found the Regulatory Role of NPM1 in Inflammatory Bowel Disease and Colorectal Cancer
 Sungen Biomedical Was Awarded the Most Potential Seed Enterprise 2024 by EY Fudan
On September 27, the award ceremony of EY Fudan Most Potential Enterprise 2024 and the seminar on high-growth enterprises were held in Shanghai.Beijing Sungen Biomedical Technology Co., Ltd. (Sungen B...
Sungen Biomedical Was Awarded the Most Potential Seed Enterprise 2024 by EY Fudan
On September 27, the award ceremony of EY Fudan Most Potential Enterprise 2024 and the seminar on high-growth enterprises were held in Shanghai.Beijing Sungen Biomedical Technology Co., Ltd. (Sungen B...